 Like most tools, constant use and exposure tends to make one blase about the inherent and intellectual beauty. "To a chemist, the periodic table is a tool and Leitmotif in the same way that word and letters are the trade of the literary world. "It is an essential item on every chemist's bookshelf." - Foundations of Chemistry, Vol 9, 2007 "A book that is truly the definitive work in its field: The Periodic Table by Scerri." - Foundations of Chemistry, Vol 9, 2007 ".the quality is not merely skin deep, there is a real scholarship inside.I would have been proud to have written this book rather than just contributing one image." - Gordon Woods, Education in Chemistry, "This is undoubtedly a book that every practising chemist and chemistry educator should read because of its far-reaching implications for understanding the nature of the periodic law and the challenges it presents to contemporary portrayals of the Periodic Table." - Kevin Berg, Newsletter of International History, Philosophy and Science Teaching Group "Strangely, relatively few books have been devoted to it, which makes Scerri's particularly welcome - all the more so since not only does he recount events leading up to its discovery, but also analyses its underlying meaning and implications." - John Emsley, TLS By writing this book Scerri has done us a significant service." - Chemical Educator, Volume 12. It will also prove valuable for those who teach chemistry. This book is essential reading for any school chemistry teacher and is recommended for college or university chemistry lecturers." - Metascience (2008) 17:155-157 "Eric Scerri's first book is timely, fluently written, and full of interesting ideas. He has broadened our minds." - Struc Chem 2008 We need not agree with every conclusion he draws, but his ideas will certainly set us thinking, which, of course, is what good science is all about. By writing this book and describing his philosophy, Scerri has done us a significant service. "Every chemist educator should read this book. ".valuable for students and teachers in sciences, as well as in the philosophy, and any other discipline that has some reference to chemistry." - Ivan Juranić JSCS The book closes with an examination of further chemical aspects including lesser known trends within the periodic system such as the knight's move relationship and secondary periodicity, as well at attempts to explain such trends. Finally, chapter 10 considers the way that the elements evolved following the Big Bang and in the interior of stars. Chapter 8 discusses the response to the new physical theories by chemists such as Lewis and Bury who were able to draw on detailed chemical knowledge to correct some of the early electronic configurations published by Bohr and others.Ĭhapter 9 provides a critical analysis of the extent to which modern quantum mechanics is, or is not, able to explain the periodic system from first principles. Chapters 6 and 7 consider the impact of physics including the discoveries of radioactivity and isotopy and successive theories of the electron including Bohr's quantum theoretical approach. Two chapters are devoted to the discoveries of Mendeleev, the leading discoverer, including his predictions of new elements and his accommodation of already existing elements. In chapter 3 the discovery of the periodic system by six independent scientists is examined in detail. The precursors to the periodic system, like Döbereiner and Gmelin, are discussed. The book then turns to a systematic account of the early developments that led to the classification of the elements including the work of Lavoisier, Boyle and Dalton and Cannizzaro. The Periodic Table begins with an overview of the importance of the periodic table and of the elements and it examines the manner in which the term 'element' has been interpreted by chemists and philosophers. 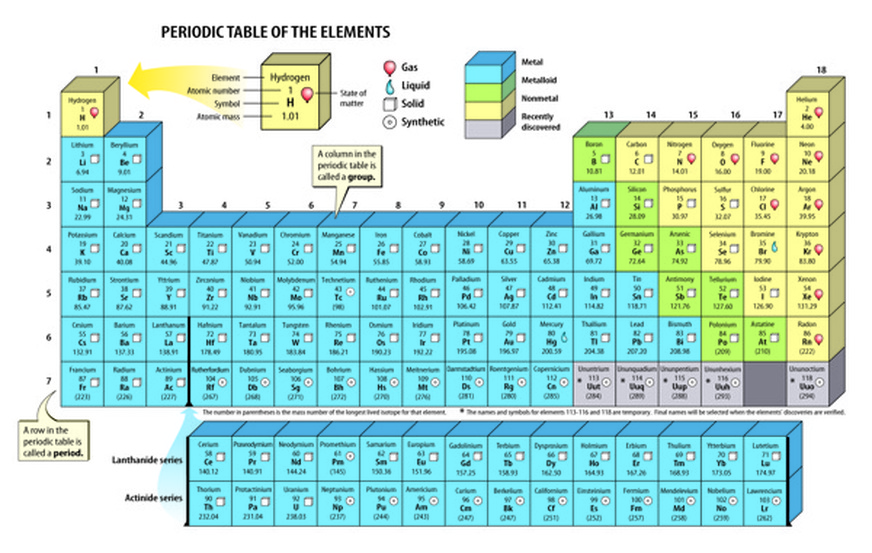
The book is written in a lively style to appeal to experts and interested lay-persons alike. 
The present book provides a successor to van Spronsen, but goes further in giving an evaluation of the extent to which modern physics has, or has not, explained the periodic system. 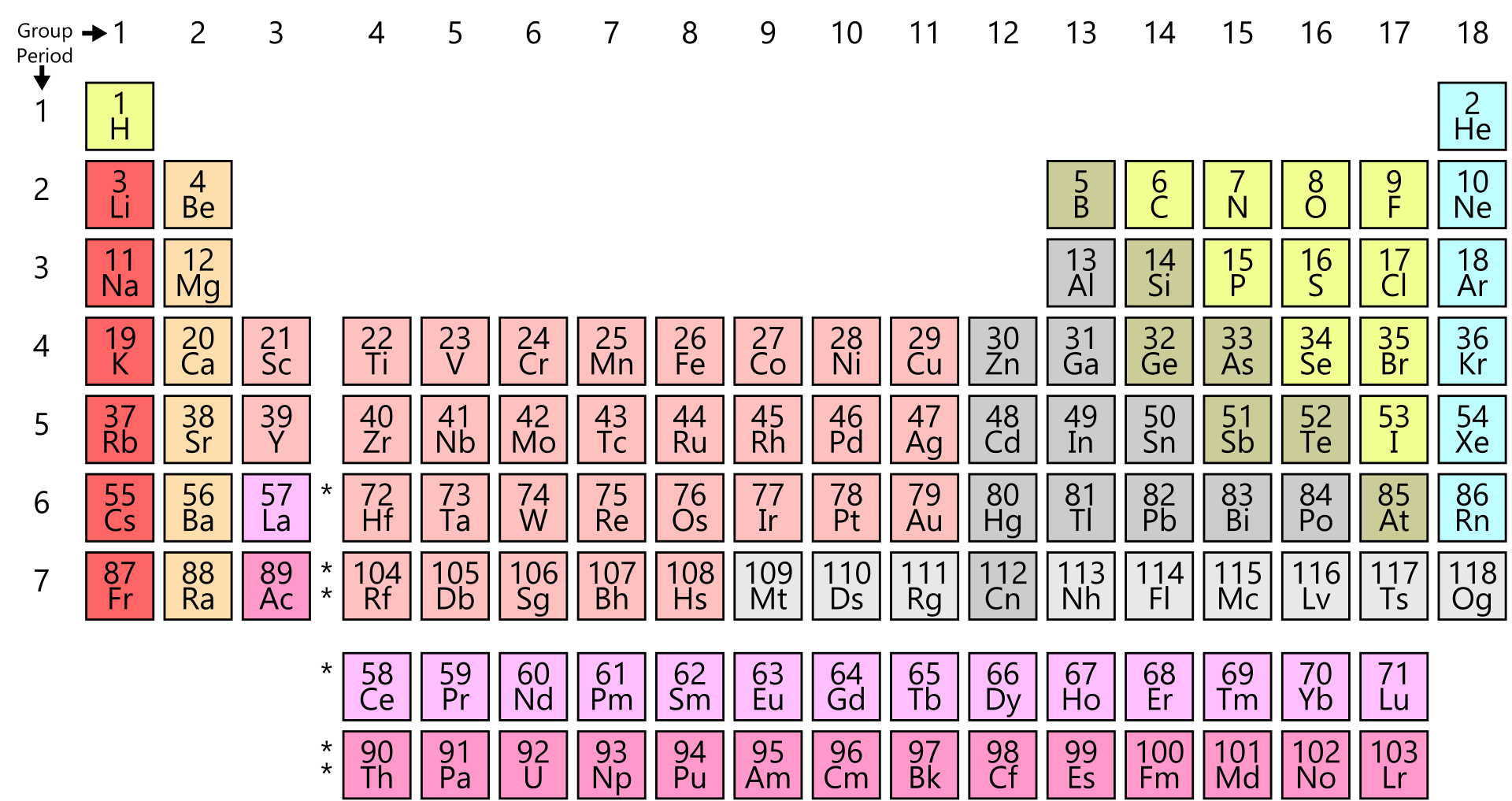
The one definitive text on the development of the periodic table by van Spronsen (1969), has been out of print for a considerable time. 
It lies at the core of chemistry and embodies the most fundamental principles of the field. The periodic table is one of the most potent icons in science.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
